UbiProetin, where recombinant technology meets scientific excellence
Product
Product Overview


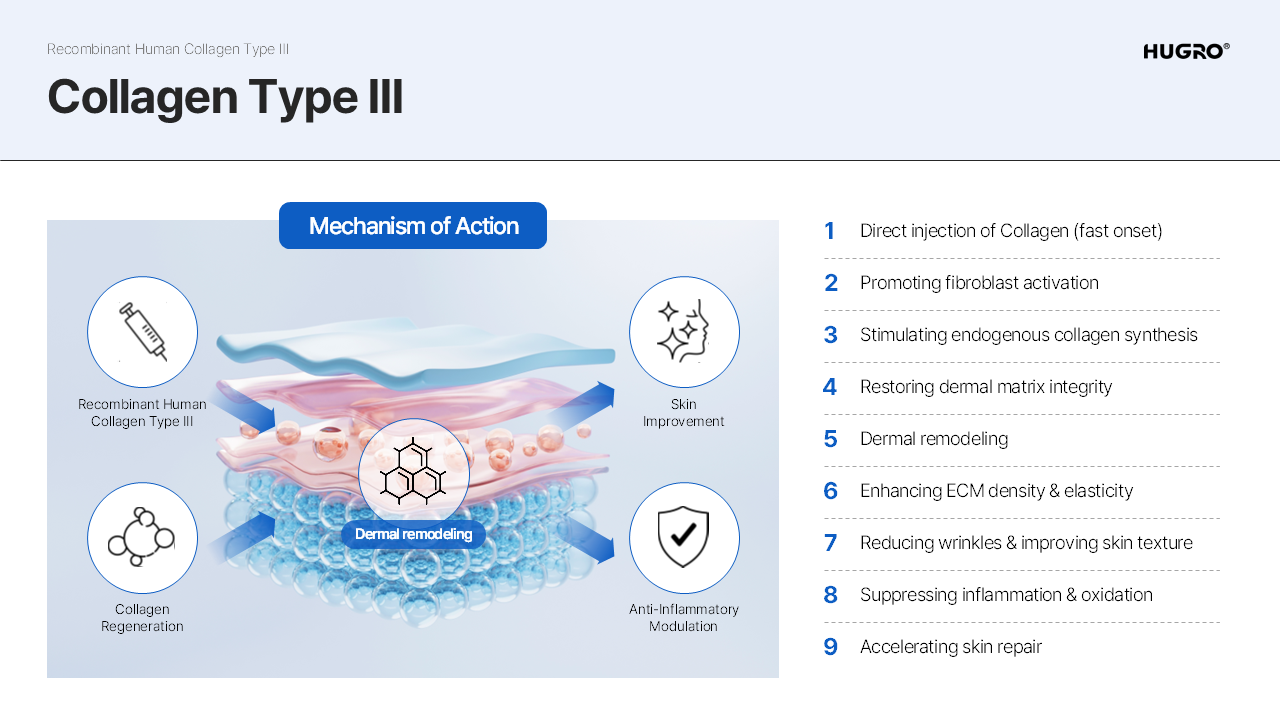
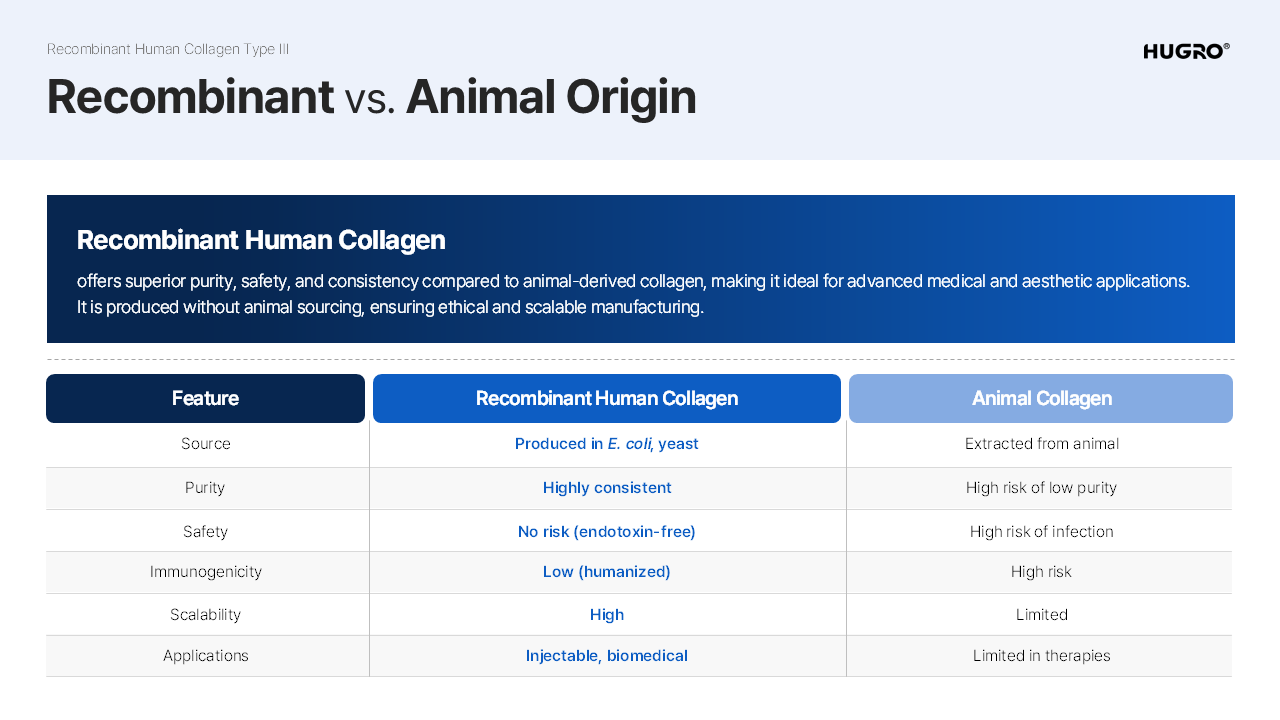
strength
Data
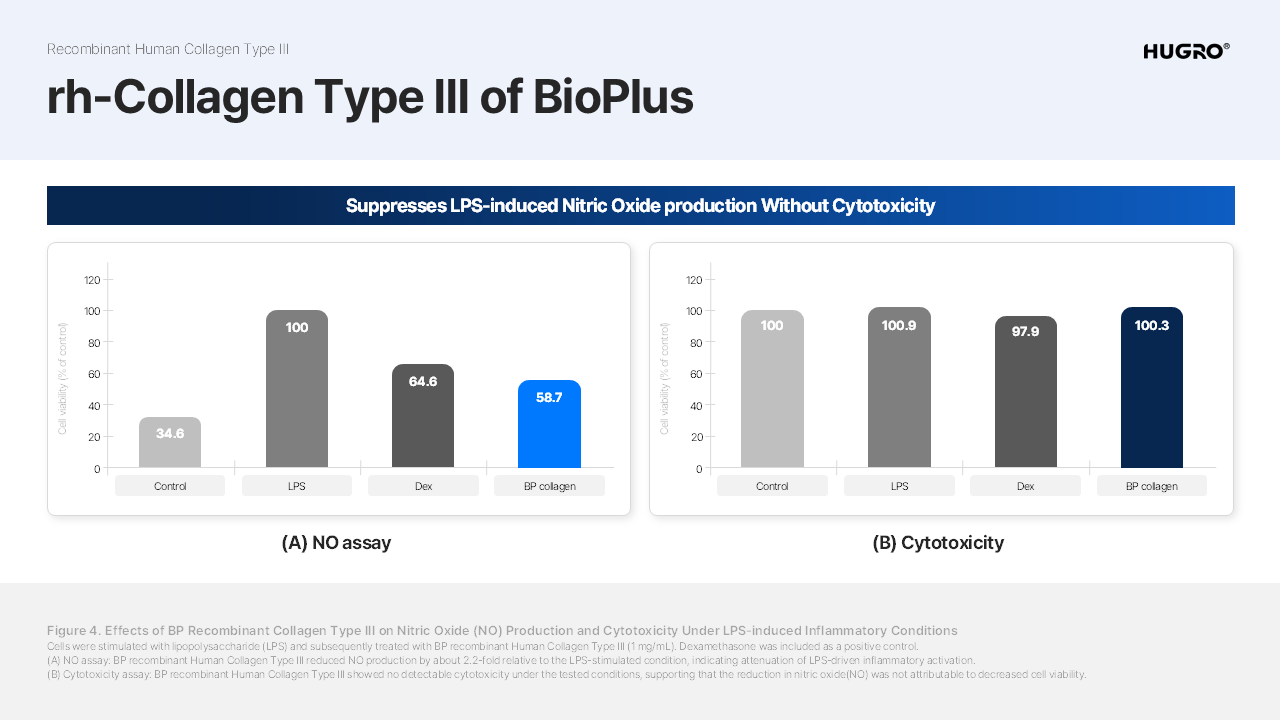
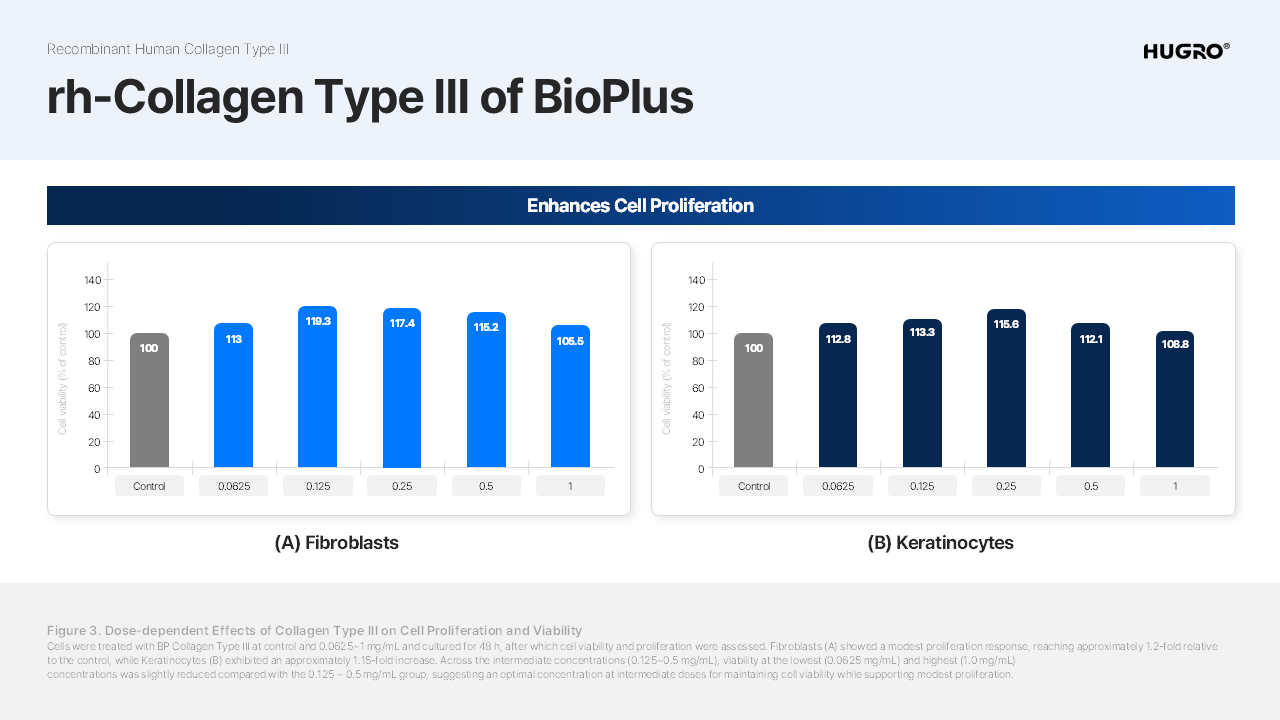
Product Details
| Product Specifications | |
|---|---|
| Description | Steriled, Vial, Mimicking human dermal matrix (Collagen 90%, Elastin 10%) |
| Appearance | Lyophilized powder, white |
| Solubility | Should be soluble in an aqueous solution |
| pH | 7.0 ~ 8.0 |
| MW | 90 kDa (Triple Helix) |
| Protein content | ≥95% |
| Water | ≤10% |
| Ignited residue | ≤2.0% |
| Collagen purity | ≥98% |
| Heavy metal (e.g. Pb) | ≤10 ug/g |
| As, Hg, Cd, Cr ≤ 1ug/g | |
| Cu, Mo, Fe, and Ni ≤ 50ug/g | |
| Total number of aerobic bacteria | ≤10 CFU/g |
| Mold & Yeast | ≤10 CFU/g |
| Staphylococcus aureus | Negative |
| Pseudomonas aeruginosa | Negative |
| Escherichia coli | Negative |
| Antibiotic residue | Should not have kanamicin activity |
| Resiamount of exogenous DNA | **≤10 ng/mg |
| Odor | Odorless or with slight characteristic odor |
| Identification 1 | FT-IR |
| Infrared spectrum | Consistent with reference spectrum |
| Osmolality | 250 - 350 mOsmol/kg |
| Thermal stability | No gel or floc appears at 57±0.5°C for 4h |
| Bacterial endotoxins | ≤0.05 EU/mg |
| Host Cell Protein (HCP) | **≤10 ng/mg |
| Expiration data | 3 year from date of manufacture |



